Question 7: Write a comprehensive note on nuclear fission.
ANSWER
Nuclear Fission
It is the nuclear reaction in which a heavy nucleus splits up into two nuclei and a huge amount of energy is released.
In 1939, Hann and Stressmann were making similar experiments as Fermi did in 1934. However, they found that the product nuclei were not of some heavier element (Z>92) but Barium and Krypton. Hann and Stressmann were the first who observed the nuclear fission reaction.
The reaction can be explained as when the thermal neutron (of energy 0.025 eV, and this energy corresponds to a neutron moving with a velocity of 2000 m/s) is bombarded on target nucleus of 92U235.
For such a nuclear reaction, neutron is far better than using α-particles or γ-rays. It is because neutron is chargeless and does not experience any Coulomb force while approaching the nucleus. Therefore, it penetrates very far in the nucleus.
When neutron is incident on the target nucleus, the nucleus is broken apart in two or more fragments of moderate sizes.
0n1+ 92U235 →92U236* → 36Kr92 + 56Ba141+ 30n1 + Q
… (A)
It was observed that there is a decrease in the mass of the system and hence some mass converts as the released energy. The amount of energy released in the reaction is of high amount. Moreover, three neutrons are emitted in the process. Calculation shows that when one atom of 92U235 is disintegrated, 200 Mev of energy is released.
Fission Chain
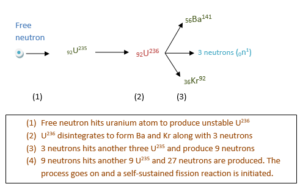
The release of three neutrons in the fission reaction can be utilized to cause further fission reactions. With every such reaction, number of neutrons released will increase which will further continue and increase the fission. In this way a self-sustaining fission reaction is set up with a single neutron. Such a self-sustaining reaction is called Fission Reaction.
So when such a reaction continues, the number of neutrons would increase and whole of 92U235 would be transformed with the release of an enormous amount of energy. The huge amount of energy can cause a violent explosion with a huge destruction. This is the principle of atomic bomb. Therefore, a controlled smooth reaction is necessary for constructive purposes.
The condition are made in such a way that the increased number of neutrons released are allowed to escape or captured by some non-fissionable substance.
For a smooth fission reaction we need a minimum amount of Uranium. However, if the amount of Uranium is less than the small amount, the fission reaction will die soon. Therefore, it is necessary that the mass of Uranium must be greater than a minimum mass. The minimum mass of Uranium required to set up a smooth fission
reaction is called critical mass.
Therefore, due to the release of huge amount of energy, fission reaction can be utilized for the production of tremendous energy for peaceful and constructive purposes

Pingback:nuclear-reactor-construction-principle-working-theory-and-uses – msa
Pingback:Definition and explanation of nuclear-reactions – msa
Pingback:long-questions-ch20-p12 – msa