Generally, if there are N
0 number of the radioactive atoms of the material at a certain time, t = 0, then
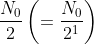 number of atoms will decay in the first half-life and N0/2 number will survive.
number of atoms will decay in the first half-life and N0/2 number will survive.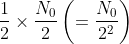 of atoms will decay in the second half-life and the same number will survive.
of atoms will decay in the second half-life and the same number will survive.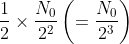 number of atoms will decay in the third half-life and the same number will survive.
number of atoms will decay in the third half-life and the same number will survive.
Generally, after n half-lifes the number that decay (or survive) is
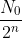
Pingback:Definition and explanation of nuclear-reactions – msa
Pingback:binding-energy-and-mass-defect – msa
Pingback:long-questions-ch20-p12 – msa