Question 15: Work can be converted completely into heat, so can heat be converted completely into work?
ANSWER
Work can be completely converted to heat. Producing heat by rubbing two surfaces (for example, rubbing hands in cold weather to warm them) is a good example of converting work into heat. Similarly when brakes are applied to stop a moving car, the kinetic energy is converted to heat (rubbing tyres with the road and disks of the car). In all such cases kinetic energy is completely converted to heat. Joule, in 1845, made a series of experiments establishing that work can be completely converted into heat. He showed by experiments that W = JQ where J is Joule constant and in SI units J= 1.
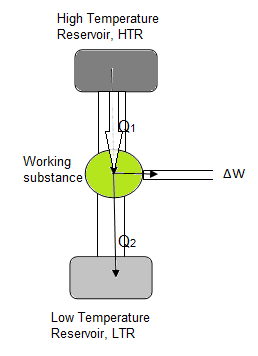
But can heat be completely converted into work? The answer is”no”. The second law of thermodynamics states that the conversion of heat to work is never 100% efficient; some of the heat must be lost! A heat engine which converts heat to other forms of energy (including work) is schematically pictured below.
The important aspect of the engine is that some heat must be transferred to the low temperature reservoir, and no heat engine can work without this LTR. The efficiency of a heat engine is given by, % efficiency = (1 – T2/T1)x 100
This efficiency will be 100% if, and only if, T2 = 0
But T2 can not be zero by the second law of thermodynamics, because the LTR must be there. So, heat energy can’t be completely converted to work.

Pingback:index-s10-p11 – msa